A Healthy Glass of Water Against the Silent Killer
“We are trapped between kidney diseases and day-to-day income”
A common yet despairing phrase among individuals, particularly in North Central Sri Lanka. Some of them might not know that the water they drink is contaminated with inorganic species such as heavy metals and fluoride, which is considered hazardous in many parts of the world. Chronic kidney disease with unknown etiology (CKDu), has become the silent killer that has perplexed the medical community.
Even though its cause remains unknown, it is correlated with the intake of water contaminated with F–, Pb(II), Cd(II), and As(V). With many materials already developed to remove both organic and inorganic contaminants from water, a high concern lies in their cost and their action as secondary pollutants. Hence the need for cost-effective, nontoxic, and biodegradable adsorbents which can effectively adsorb several contaminants simultaneously and remove them rapidly, remained at large over the past years.
Until recently, a group of dedicated scientists was able to develop a promising solution by conducting a research at the Centre for Advanced Materials and Devices (CAMD), Department of Chemistry, University of Colombo. That is the “Biopolymer-Based Nanohydroxyapatite Composites for the Removal of Fluoride, Lead, Cadmium, and Arsenic from Water”. This publication was published on March 18th, 2021, in “ACS Omega” which is one of the prestigious journals that publishes high-quality new findings. With Prof. Rohini M de Silva from the University of Colombo as the corresponding author, along with the rest of the authors mentioned below, was successful in presenting this innovation by harnessing the essence of central science.

Prof. Rohini M. de Silva
Department of Chemistry, Faculty of Science,
University of Colombo

Senior Prof. K. M. Nalin de Silva
Department of Chemistry, Faculty of Science, University of Colombo

Senior Prof. D. P. Dissanayake
Department of Chemistry, Faculty of Science, University of Colombo

Mrs. M. Shanika Fernando
Department of Chemistry, Faculty of Science, University of Colombo

Mrs. A. K. D. V. K. Wimalasiri
Department of Chemistry, Faculty of Science, University of Colombo

Prof. K. R. Koswattage
Faculty of Technology, Sabaragamuwa University

Dr. Karolina Dziemidowicz
UCL School of Pharmacy,
University College London

Prof. Gareth R. Williams
UCL School of Pharmacy,
University College London
The use of Nano-hydroxyapatite (HAP) and its composites have shown effective adsorption of water pollutants due to its low toxicity, biocompatibility, and ability to show versatile sorption properties toward both anions and cations. This study focused on overcoming the challenges of using nanoparticles alone as sorbents, that is by combining HAP with natural biopolymers. Furthermore, it addressed the lack of understanding on how the type of polymer incorporated and their component ratios affect the sorption properties of HAP biopolymer composites. Hence the HAP nanocomposites were prepared with 4 biopolymers namely, Chitosan (HAP-CTS), Carboxymethyl Cellulose (HAP-CMC), Alginate (HAP-ALG) and Gelatin (HAP-GEL).
The nanocomposites with optimal HAP to biopolymer ratios were synthesized using a simple wet chemical in situ precipitation method. The successful synthesis of the composites was revealed with the characterization using advanced techniques such as scanning electron microscopy, Fourier transform infrared spectroscopy, X-ray diffraction, Brunaeur-Emmett-Teller surface area analysis, and thermogravimetric analysis.
The adsorption abilities of the above materials toward Pb(II), Cd(II), As(V), and F– were investigated under adsorption studies conducted between pH range of 3 to 11 for an incubation time of 1 hour. Out of the four nanocomposites, HAP-CTS showed the best adsorption properties towards all ions examined, and was identified as the most versatile adsorbent which works over a wide range of pH values, including at neutral pH.
This is related to the presence of NH2 in chitosan (CTS), its cationic nature compared to the other three polymers, and high surface area. Based on the observations made in the adsorption studies along with elemental mapping and XPS data, a mechanism for the adsorption of Multi ions onto HAP-CTS was put forward.
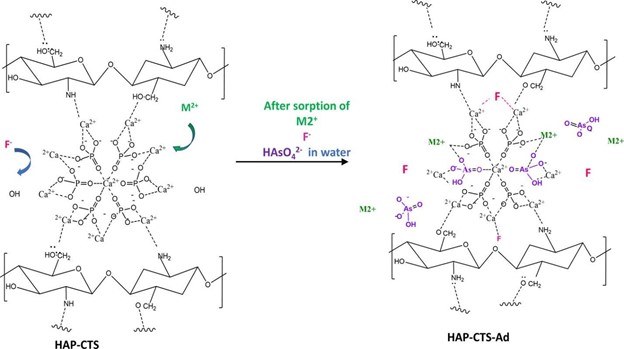
As shown above, the negatively charged ions (F– and AsO3-4) get adsorbed either through an exchange process with OH– or by adhering to Ca2+ sites. The cations Pb(II) and Cd(II) mainly undergo ion exchange with Ca2+ as well as get immobilized by surface interactions with the negatively charged groups of the HAP-CTS structure.
With HAP-CTS as the best polymer composite in hand, it was then integrated with other matrices to develop devices for water purification as the powdered form is not generally favorable in real applications as filters. Therefore, HAP-CTS was further modified by integrating cotton gauze (CG) and granular activated carbon (GAC) to form the resultant materials named HAP-CTS-CG and HAP-CTS-GAC respectively.
Gravitation filtration studies were conducted for HAP-CTS powder, HAP-CTS-GAC and HAP-CTS-GC incorporating the real-life concentrations of the mixture of ions with the prevalent concentration in the groundwater of Sri Lanka and the maximum levels reported in the CKDu prominent areas. It had shown that the powdered form of HAP-CTS is the best sorbent giving the highest breakthrough capacities of 3000, 3000, 2699, and 2000 mL/g for Pb(II), Cd(II), As(V), and F– respectively. The breakthrough capacities were calculated considering the safe limits for the said ions according to the WHO standards.
It has been estimated that the incidence of CKDu in some parts of Sri Lanka has been doubling every four to five years. In the silent wait for survival, people affected by this disease are calling for research so that others can be saved from the same fate.
This committed group of scientists has answered their call by addressing this medical mystery by providing a promising solution with the use of Biopolymer-Based Nanohydroxyapatite Composites in water purification. The research team gratefully acknowledges the National Research Council Sri Lanka for the financial support and the academic and technical staff of the Department of Chemistry, Faculty of Science, University of Colombo for their immense dedication.
Reference:
M. Shanika Fernando, A. K. D. V. K. Wimalasiri, Karolina Dziemidowicz, Gareth R. Williams, K. R. Koswattage, D. P. Dissanayake, K. M. Nalin De Silva, And Rohini M. De Silva*. (2021, March 18). Biopolymer-Based Nanohydroxyapatite Composites for the Removal of Fluoride, Lead, Cadmium, and Arsenic from Water. ACS Omega 2021, 6(12), 8517–8530. https://pubs.acs.org/doi/10.1021/acsomega.1c00316?ref=pdf
Title Image Credits:
● https://bit.ly/2Z8xDI5
● https://bit.ly/3igYVCT
● https://bit.ly/3kvFM1P
Written by: K. V. Dilshan T. Chandraguptha